2 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
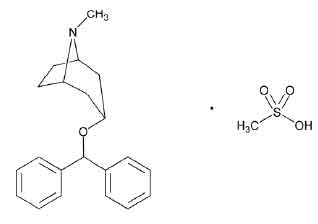
BENZTROPINE MESYLATE TABLETS USP (Benztropine Mesylate) is a synthetic compound containing structural feature found in atropine and diphenhydramine. Chemically, Benztropine Mesylate is designated as 8-Azabicyclo[3.2.1]octane, 3-(diphenylmethoxy)-N -methyl-, endo -, methanesulfonate. Its molecular formula is C21H25NO.CH4O3S and its molecular weight is 403.53.
STRUCTURAL FORMULA :
Its structural formula is :
BENZTROPINE MESYLATE TABLETS USP are white coloured, circular, biconvex tablets with break line on one side and “SGP” embossed on the other side.
COMPOSITION :
Each uncoated tablet contains :
Benztropine Mesylate USP 2 mg
Excipients q.s.
ACTIONS :
Benztropine is a centrally acting anticholinergic agent with antihistaminic properties resulting from the combination of the tropine portion of the atropine molecule and the benzohydryl portion of diphenhydramine. Animal studies have indicated that anticholinergic activity of benztropine is approximately half that of atropine, while antihistaminic activity approaches that of pyrilamine. Its anticholinergic effects have been established as therapeutically significant in the management of parkinsonism. Benztropine antagonises the effect of acetylcholine, decreasing the imbalance between the neurotransmitters acetylcholine and dopamine, which may improve the symptoms of early parkinson’s disease.
PHARMACOKINETICS :
Benztropine is absorbed from the GI tract, crosses the blood-brain-barrier, and may cross the placenta. After oral administration, a small part of the dose may pass through the GI tract unchanged into the faeces. It has been reported that the onset of action of benztropine is
1 to 2 hours and duration of action is upto 24 hours. It binds extensively, approximately 95 %, with serum proteins. The metabolism of benztropine is unknown, but most of the drug is excreted renally, both as a parent drug and as metabolites.
INDICATIONS :
BENZTROPINE MESYLATE TABLETS USP is recommended for all forms of parkinsonism including arteriosclerotic, postencephalitic, idiopathic, as well as medicine-induced extrapyramidal disorders (except tardive dyskinesia). It can be effective at any stage of the disease, even when a patient has become bedridden. BENZTROPINE MESYLATE TABLETS USP often is helpful in patients who have become unresponsive to other agents. BENZTROPINE MESYLATE TABLETS USP is a powerful anticholinergic agent which is mainly effective in relieving tremor and rigidity. Therapy is directed toward control of disturbing symptoms to permit the patient maximum integration of function with minimum discomfort. In non-medicine-induced parkinsonism, partial control of symptoms is usually achieved.
Administration :
BENZTROPINE MESYLATE TABLETS USP is for oral administration.
Dosage :
Because of cumulative action of BENZTROPINE MESYLATE TABLETS USP, therapy should be initiated with a low dose which is increased gradually at five or six-day intervals to the smallest amount necessary for optimal relief. Increases should be made in increments of 0.5 mg to a maximum of 6 mg or until optimal results are obtained without excessive adverse reactions.
Postencephalitic and Idiopathic Parkinsonism :
The usual daily dose is 1 to 2 mg, with a range of 0.5 to 6 mg orally or parenterally. As with any agent used in parkinsonism, dosage must be individualized according to age and weight, and the type of parkinsonism being treated. Generally, older patients and thin patients cannot tolerate large doses. Most patients with postencephalitic parkinsonism need fairly large doses and tolerate them well. Patients with a poor mental outlook are usually poor candidates for therapy. In idiopathic parkinsonism, therapy may be initiated with a
single daily dose of 0.5 to 1 mg at bedtime. In some patients, this will be adequate; in others 4 to 6 mg a day may be required. In postencephalitic parkinsonism, therapy may be initiated in most patients with 2 mg a day in one or more doses. In highly sensitive patients, therapy may be initiated with 0.5 mg at bedtime, and increased as necessary.
Some patients experience greatest relief by taking the entire dose at bedtime; others react more favourably to divided doses, two to four times a day. Frequently, one dose a day is sufficient and divided doses may be unnecessary or undesirable. The long duration of action of this drug makes it particularly suitable for bedtime medication when its effects may last throughout the night, enabling patients to turn in bed during the night more easily and to rise in the morning. When BENZTROPINE MESYLATE TABLETS USP is started, do not terminate therapy with other antiparkinsonian agents abruptly. If the other agents are to be reduced or discontinued, it must be done gradually. Many patients obtain greatest relief with combination therapy. BENZTROPINE MESYLATE TABLETS USP may be used concomitantly with Carbidopa-Levodopa Tablets, or with Levodopa tablets, in which case periodic dosage adjustment may be required in order to maintain optimum response.
Drug - Induced Extrapyramidal Disorders :
In treating extrapyramidal disorders due to neuroleptic drugs (e.g., phenothiazines), the recommended dosage is 1 to 4 mg once or twice a day orally or parenterally. Dosage must be individualized according to the need of the patient. Some patients require more than recommended; others do not need as much. In acute dystonic reactions, 1 to 2 ml of the injection usually relieves the condition quickly. After that, the tablets, 1 to 2 mg twice a day, usually prevent recurrence. When extrapyramidal disorders develop soon after initiation of treatment with neuroleptic drug (e.g., phenothiazines), they are likely to be transient. 1 to 2 mg of BENZTROPINE MESYLATE TABLETS USP two or three times a day usually provides relief within one or two days. After one or two weeks, the drug should be withdrawn to determine the continued need for it. If such disorders recur, BENZTROPINE MESYLATE TABLETS USP can be reinstituted. Certain drug - induced extrapyramidal disorders that develop slowly may not respond to BENZTROPINE MESYLATE TABLETS USP.
CONTRAINDICATIONS :
BENZTROPINE MESYLATE TABLETS USP is contraindicated in angleclosure glaucoma; myasthenia gravis; pyloric or duodenal obstruction; stenosing peptic ulcer; prostatic hypertrophy or bladder neck obstructions; megacolon; tardive dyskinesia; children less than 3 years old. BENZTROPINE MESYLATE TABLETS USP is contraindicated in patients who are hypersensitive to any component in this product. BENZTROPINE MESYLATE TABLETS USP contains lactose which is contraindicated in patients with galactosaemia, the glucose-galactose malabsorption syndrome, or lactase deficiency.
When BENZTROPINE MESYLATE TABLETS USP is given concomitantly with phenothiazines, haloperidol, or other drugs with anticholinergic or antidopaminergic activity, patients should be advised to report gastrointestinal complaints, fever or heat intolerance
promptly. Paralytic ileus, hyperthermia and heat stroke, all of which have sometimes been fatal, have occurred in patients taking anticholinergic-type antiparkinsonism drugs, including BENZTROPINE MESYLATE TABLETS USP, in combination with phenothiazines and/or tricyclic antidepressants. Since BENZTROPINE MESYLATE TABLETS USP contains structural features of atropine, it may produce anhidrosis. For this reason, it should be administered with caution during hot weather, especially when given concomitantly with other atropine-like drugs to the chronically ill, the alcoholic, those who have central nervous system disease and those who do manual labour in a hot environment. Anhidrosis may occur more readily when some disturbance of sweating already exists. If there is evidence of anhidrosis, the possibility of hyperthermia should be considered. Dosage should be decreased at the discretion of the physician so that the ability to maintain body heat equilibrium by perspiration is not impaired. Severe anhidrosis and fatal hyperthermia have occurred.
PRECAUTIONS :
General :
Since BENZTROPINE MESYLATE TABLETS USP has cumulative action, continued supervision is advisable. Patients with a tendency to tachycardia and patients with prostatic hypertrophy should be observed closely during treatment. Dysuria may occur, but rarely becomes a problem. Urinary retention has been reported with BENZTROPINE MESYLATE TABLETS USP. The drug may cause complaints of weakness and inability to move particular muscle groups, especially in large doses. For example, if the neck has been rigid and suddenly relaxes, it may feel weak, causing some concern. In the event, dosage adjustment is required. Mental confusion and excitement may occur with large doses, or in susceptible patients. Visual hallucinations have been reported occasionally. Furthermore, in the treatment of extrapyramidal disorders due to neuroleptic drugs (e.g. phenothiazines), in patients with mental disorders, occasionally there may be intensification of mental symptoms. In such cases, antiparkinsonian drugs can precipitate a toxic psychosis. Patients with mental disorders should be kept under careful observation, especially at the beginning of treatment or if dosage is increased.
Tardive dyskinesia may appear in some patients on long-term therapy with phenothiazines and related agents, or may occur after therapy with these drugs has been discontinued. Antiparkinsonism agents do not alleviate the symptoms of tardive dyskinesia and in some instances may aggravate them. BENZTROPINE MESYLATE TABLETS USP is not recommended for use in patients with tardive dyskinesia. The physician should be aware of the possible occurrence of glaucoma. Although the drug does not appear to have any adverse effect on simple glaucoma, it probably should not be used in angle-closure glaucoma. BENZTROPINE MESYLATE TABLETS USP should also be used with caution in patients with urinary retention, cardiovascular disease and hepatic or renal impairment. BENZTROPINE MESYLATE TABLETS USP should be used cautiously in diabetic patients.
Pregnancy : Category C
It is not known whether BENZTROPINE MESYLATE TABLETS USP can cause foetal harm when administered to a pregnant woman or can affect reproductive capacity. BENZTROPINE MESYLATE TABLETS USP should be given to a pregnant woman only if clearly needed.
Nursing Mothers :
It is not known whether this medicine is excreted in human milk. Because many medicines are excreted in human milk, caution should be exercised when BENZTROPINE MESYLATE TABLETS USP is administered to a nursing mother.
Paediatric Use :
Because of the atropine-like side effects, BENZTROPINE MESYLATE TABLETS USP should be used with caution in paediatric patients over three years of age.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
BENZTROPINE MESYLATE TABLETS USP may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle.
INTERACTIONS :
When BENZTROPINE MESYLATE TABLETS USP is given concomitantly with anticholinergics or those with antidopaminergic activity, such as phenothiazines, haloperidol or other such drugs, patients should be advised to report fever, heat intolerance and gastrointestinal complaints promptly. Paralytic ileus, sometimes fatal, has occurred in patients taking anticholinergic-type antiparkinsonism drugs, including BENZTROPINE MESYLATE TABLETS USP, in combination with phenothiazines and/or tricyclic antidepressants.
Alcohol and other CNS depressants, such as anxiolytics, sedatives and hypnotics, can increase the sedative effects of benztropine. Drugs that exert anticholinergic properties may pharmacodynamically oppose the effects of prokinetic agents such as cisapride or metoclopramide. The doses of BENZTROPINE MESYLATE TABLETS USP and levodopa must be adjusted when the drugs are given simultaneously. Through its central anticholinergic actions BENZTROPINE MESYLATE TABLETS USP can potentate the dopaminergic effects of levodopa. While some patients may benefit from this interaction, clinicians should be ready to decrease doses of levodopa if benztropine is added. The anticholinergic properties of BENZTROPINE MESYLATE TABLETS USP, by slowing GI transit, may decrease levodopa bioavailability. However, this mechanism appears to be of modest clinical significance. Anticholinergics can raise intragastric pH. This effect may interfere with the oral bioavailability of
metoconazole. BENZTROPINE MESYLATE TABLETS USP should be used cautiously in patients receiving ketoconazole. Opiate agonists should be used cautiously with anticholinergics since additive depressive effects on GI motility or bladder function may be seen. The muscarinic actions of drugs known as parasympathomimetics, including both direct cholinergic receptor agonists and cholinesterase inhibitors, can antagonise the anticholinergic actions of benztropine. Benztropine might also antagonise some of the effects of the parasympathomimetics. Carbonic anhydrase inhibitors increase the alkalinity of the urine, thereby increasing the amount of nonionized drug available for renal tubular reabsorption. Use with caution if BENZTROPINE MESYLATE TABLETS USP is administered with carbonic anhydrase inhibitors, which can decrease excretion and enhance the effects of BENZTROPINE MESYLATE TABLETS USP. Monitor for excessive anticholinergic adverse effects.
SIDE EFFECTS :
Adverse effects, most of which are anticholinergic or antihistaminic in nature are listed below by body system in order of decreasing severity.
Cardiovascular : Tachycardia.
Digestive : Constipation, dry mouth, nausea, vomiting, paralytic ileus. If dry mouth is so severe that there is difficulty in swallowing or speaking, or loss of appetite and weight occur, reduce dosage, or discontinue the medicine temporarily. Slight reduction in dosage may control nausea and still give sufficient relief of symptoms. Vomiting may be controlled by temporary discontinuation, followed by resumption at a lower dosage.
Nervous System : Toxic psychosis, including confusion, disorientation, memory impairment, visual hallucinations, exacerbation of pre-existing psychotic symptoms; nervousness; depression; listlessness; numbness of fingers.
Special Senses : Blurred vision, dilated pupils.
Urogenital : Urinary retention, dysuria.
Metabolic/Immune and Skin : Occasionally, an allergic reaction, e.g. skin rash, develops. If this cannot be controlled by dosage reduction, the medication should be discontinued.
Other : Heat stroke, hyperthermia, fever.
OVERDOSAGE :
Manifestations :
As with any of those seen in atropine poisoning or antihistamine overdosage : CNS depression, preceded or followed by stimulation; confusion; nervousness; listlessness; intensification of mental symptoms or toxic psychosis in patients with mental illness being treated with phenothiazine derivatives; hallucination (especially visual); dizziness; muscle weakness; ataxia; dry mouth; mydriasis; blurred vision; palpitations; tachycardia; nausea; vomiting; dysuria; numbness of fingers; dysphagia; allergic reactions, e.g. skin rash; headache; hot, dry, flushed skin, delirium; coma; shock; convulsions; respiratory arrest; anhydrosis; hyperthermia; glaucoma; constipation.
TREATMENT OF OVERDOSAGE :
Physostigmine salicylate, 1 to 2 mg SC or IV, will reverse symptoms of anti-cholinergic intoxication. A second injection may be given after 2 hours if required. Otherwise treatment is symptomatic and supportive. Induce emesis or perform gastric lavage (contraindicated in precomatose, convulsive, or psychotic states). Maintain respiration. A short-acting barbiturate may be used for CNS excitement, but with caution to avoid subsequent depression; supportive care for depression (avoid convulsant stimulants such as picrotoxin,
pentylenetetrazol, or bemegride); artificial respiration for severe respiratory depression; a local miotic for mydriasis and cycloplegia; ice bags or other cold applications and alcohol sponges for hyperpyrexia, a vasopressor and fluids for circulatory collapse. Darken room for photophobia.
STORAGE :
Store below 30°C (86°F), protected from moisture and light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
BENZTROPINE MESYLATE TABLETS USP contains Benztropine Mesylate USP 2 mg.
10 Blisters of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.
 Cardiovascular
Cardiovascular