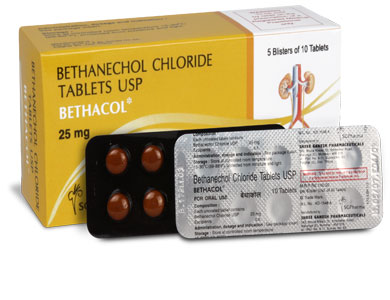
25 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
BETHACOL (Bethanechol Chloride), a cholinergic agent, is a synthetic ester which is structurally and pharmacologically related to acetylcholine. Chemically, Bethanechol Chloride is 1-Propanaminium, 2-(aminocarbonyl)oxy-N, N, N-trimethyl-, chloride, (±)-. Its molecular formula is C7H17CIN2O2 and its molecular weight is 196.67.
STRUCTURAL FORMULA :
Its structural formula is :
-Structure.jpg)
BETHACOL 5 mg tablets are white in colour, round, beveled, scored tablets.
BETHACOL 10 mg tablets are white in colour, round, beveled, scored tablets.
BETHACOL 25 mg tablets are white in colour, round, beveled, scored tablets.
BETHACOL 50 mg tablets are yellow in colour, round, beveled, scored tablets.
COMPOSITION :
Each tablet for oral administration contains 5 mg, 10 mg, 25 mg, or 50 mg Bethanechol Chloride USP. 50 mg tablets also contain FD & C Yellow # 6 Lake.
ACTIONS :
Bethanechol Chloride acts principally by producing the effects of stimulation of the parasympathetic nervous system. It increases the tone of the detrusor urinae muscle, usually producing a contraction sufficiently strong to initiate micturition and empty the bladder. It stimulates gastric motility, increases gastric tone and often restores impaired rhythmic peristalsis. Stimulation of the parasympathetic nervous system releases acetylcholine at the nerve endings. When spontaneous stimulation is reduced and therapeutic intervention is required, acetylcholine can be given, but it is rapidly hydrolyzed by cholinesterase and its effects are transient. Bethanechol Chloride is not destroyed by cholinesterase and its effects are more prolonged than those of acetylcholine.
PHARMACOKINETICS :
Effects on the Gl and urinary tracts sometimes appear within 30 minutes after oral administration of Bethanechol Chloride, but more often 60 to 90 minutes are required to reach maximum effectiveness. Following oral administration, the usual duration of action of bethanechol is one hour, although large doses (300 to 400 mg) have been reported to produce effects for up to six hours. Subcutaneous injection produces a more intense action on bladder muscle than does oral administration of the drug. Because of the selective action of bethanechol, nicotinic symptoms of cholinergic stimulation are usually absent or minimal when orally or subcutaneously administered in therapeutic doses, while muscarinic effects are prominent. Muscarinic effects usually occur within 5 to 15 minutes after
subcutaneous injection, reach a maximum in 15 to 30 minutes, and disappear within two hours. Doses that stimulate micturition and defecation and increase peristalsis do not ordinarily stimulate ganglia or voluntary muscles. Therapeutic test doses in normal human subjects have little effect on heart rate, blood pressure or peripheral circulation. Bethanechol Chloride does not cross the blood-brain barrier because of its charged quaternary amine moiety. The metabolic rate and mode of excretion of the drug have not been elucidated.
INDICATIONS :
Bethanechol Chloride is indicated for the treatment of acute postoperative and postpartum nonobstructive (functional) urinary retention and for neurogenic atony of the urinary bladder with retention.
Dosage must be individualized, depending on the type and severity of the condition to be treated.Preferably give the drug when the stomach is empty. If taken soon after eating, nausea and vomiting may occur. The usual adult oral dose ranges from 10 to 50 mg three or
four times a day. The minimum effective dose is determined by giving 5 to 10 mg initially and repeating the same amount at hourly intervals until satisfactory response occurs or until a maximum of 50 mg has been given. The effects of the drug sometimes appear within 30 minutes and are usually maximal within 60 to 90 minutes. The drug effects persist for about one hour. If necessary, the effects of the drug can be abolished promptly by atropine.
CONTRAINDICATIONS :
Hypersensitivity to BETHACOL, hyperthyroidism, peptic ulcer, latent or active bronchial asthma, pronounced bradycardia or hypotension, vasomotor instability, coronary artery disease, epilepsy and parkinsonism. Bethanechol Chloride should not be employed when the strength or integrity of the gastrointestinal or bladder wall is in question or in the presence of mechanical obstruction; when increased muscular activity of the gastrointestinal tract or urinary bladder might prove harmful, as following recent urinary bladder surgery,
gastrointestinal resection and anastomosis or when there is possible gastrointestinal obstruction; in bladder neck obstruction, spastic gastrointestinal disturbances, acute inflammatory lesions of the gastrointestinal tract or peritonitis; or in marked vagotonia.
PRECAUTIONS :
General :
In urinary retention, if the sphincter fails to relax as bethanechol contracts the bladder, urine may be forced up the ureter into the kidney pelvis. If there is bacteriuria, this may cause reflux infection.
Information for Patients :
BETHACOL should preferably be taken one hour before or two hours after meals to avoid nausea or vomiting. Dizziness, lightheadedness or fainting may occur, especially when getting up from a lying or sitting position.
Pregnancy : Teratogenic Effects : Pregnancy Category C.
Animal reproduction studies have not been conducted with Bethanechol Chloride. It is also not known whether Bethanechol Chloride can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Bethanechol Chloride should be given to a pregnant woman only if clearly needed.
Nursing Mothers :
It is not known whether this drug is secreted in human milk. Because many drugs are secreted in human milk and because of the potential for serious adverse reactions from Bethanechol Chloride in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Paediatric use :
Safety and effectiveness in paediatric patients have not been established.
Carcinogenesis, Mutagenesis, Impairment of Fertility :
Long-term studies in animals have not been performed to evaluate the effects upon fertility, mutagenic or carcinogenic potential of Bethanechol Chloride.
INTERACTIONS :
Special care is required if this drug is given to patients receiving ganglion blocking compounds because a critical fall in blood pressure may occur. Usually, severe abdominal symptoms appear before there is such a fall in the blood pressure.
SIDE EFFECTS :
Adverse reactions are rare following oral administration of bethanechol. Adverse reactions are more likely to occur when dosage is increased.
The following adverse reactions have been observed :
Body as a Whole : malaise;
Digestive : abdominal cramps or discomfort, colicky pain, nausea and belching, diarrhoea, borborygmi, salivation;
Renal : urinary urgency;
Nervous System : headache;
Cardiovascular : a fall in blood pressure with reflex tachycardia, vasomotor response;
Skin : flushing producing a feeling of warmth, sensation of heat about the face, sweating;
Respiratory : bronchial constriction, asthmatic attacks;
Special Senses : lacrimation, miosis.
Causal Relationship Unknown : The following adverse reactions have been reported and a causal relationship to therapy with bethanechol has not been established :
Body as a Whole : hypothermia;
Nervous System : seizures.
OVERDOSAGES AND TREATMENT OF OVERDOSAGE :
Early signs of overdosage are abdominal discomfort, salivation, flushing of the skin ("hot feeling"), sweating, nausea and vomiting. Atropine Sulfate is a specific antidote. The recommended dose for adults is 0.6 mg. Repeat doses can be given every two hours, according to clinical response. The recommended dosage in infants and children up to 12 years of age is 0.01 mg/kg (to a maximum single dose of 0.4 mg) repeated every two hours as needed until the desired effect is obtained or adverse effects of atropine preclude further usage. Subcutaneous injection of atropine is preferred except in emergencies when the intravenous route may be employed. The oral LD50 of Bethanechol Chloride is 1510 mg/kg in the mouse.
STORAGE :
Store at controlled room temperature 15 - 30°C (59 - 86°F), protected from moisture and light.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
BETHACOL tablets contains Bethanechol Chloride USP 5 mg / 10 mg / 25 mg / 50 mg.
5 blisters of 10 tablets per Box and 10 blisters of 10 tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.
 Cardiovascular
Cardiovascular