250 mg/10 ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
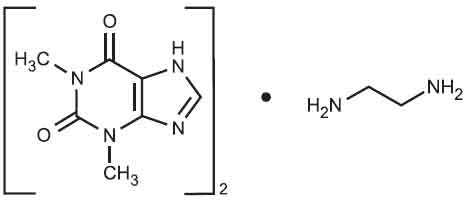
AMINOPHYLLINE INJECTION USP (Aminophylline) is used as a bronchodilator in the management of asthma and chronic obstructive pulmonary disease. Chemically, Aminophylline is 1H-Purine-2,6-dione,3,7-dihydro-1,3-dimethyl-, compd. with 1,2-ethanediamine (2:1). The molecular formula is C16H24N10O4 (anhydrous) and molecular weight is 420.43.
STRUCTURAL FORMULA :
Its structural formula is :
AMINOPHYLLINE INJECTION USP is a sterile, clear to slightly yellow colourless solution filled in ampoule of suitable size.
COMPOSITION :
Each ml contains :
Aminophylline anhydrous USP 25 mg
Water for Injection USP q.s.
Contains no preservatives.
ACTIONS :
Aminophylline is a 2:1 complex of theophylline and ethylenediamine. Aminophylline has greater water solubility than theophylline. In biological fluids aminophylline dissociates to theophylline hence the pharmacological effects of aminophylline are those of theophylline. Theophylline is a xanthine derivative with the main pharmacological action of direct relaxation of bronchial smooth muscle, relieving bronchospasm. The bronchodilatory effect of theophylline is minimal if bronchospasm is not the cause. The bronchodilatory effect may be via inhibition of selected phosophodiesterases, which produces an increase in intracellular cyclic AMP. Theophylline also directly stimulates the medullary respiratory centre. Other pharmacological effects of theophylline include stimulation of cardiac muscle (increasing both heart rate and myocardial contractility at higher doses), stimulation of the central nervous system, transient diuresis, increased gastric secretion, decreased peripheral resistance and cerebral vasoconstriction.
PHARMACOKINETICS :
Aminophylline dissociates rapidly to theophylline in biological fluids. Theophylline is rapidly distributed throughout non-adipose tissues and extracellular fluids. Theophylline crosses the placenta, and is distributed into breast milk. The concentration of theophylline in breast milk is approximately 70 % that found in the serum. The apparent volume of distribution of theophylline is 0.3 to 0.7 l/kg (average 0.45 l/kg). Approximately 60 % of theophylline in adults and 35 % in premature infants and neonates are bound to plasma proteins.
Theophylline undergoes hepatic metabolism via the cytochrome P450 system. In adults the main metabolites are 1,3-dimethyl uric acid, 1-methyl uric acid, and 3-methylxanthine. Theophylline and its metabolites undergo renal excretion.
There is significant interpatient variability in the pharmacokinetics of theophylline, and hence aminophylline. The serum half life of theophylline in otherwise healthy, non-smoking, asthmatic adults averages 7 to 9 hours, and theophylline clearance in this group is reported to be approximately 0.65 ml/kg/hr. Serum half life is increased and clearance decreased in the elderly and in patients with congestive heart failure, chronic obstructive pulmonary disease, cor pulmonale or liver disease. Serum half life is decreased and clearance increased in cigarette or marijuana smokers. Clearance in premature infants and neonates is reduced. Theophylline clearance increases during the first year of life and remains relatively constant during the first 9 years, then gradually declines to adult values by 16 years of age. Theophylline, (and hence aminophylline), has a low therapeutic index. Serum theophylline concentrations of around 5 to 20 mcg/ml (27.5 to 110 mcm/l) are generally considered therapeutic. Serum theophylline concentrations greater than 20 mcg/ml (110 mcm/l) are often associated with adverse reactions.
INDICATIONS :
AMINOPHYLLINE INJECTION USP is indicated for the treatment of reversible bronchospasm associated with chronic bronchitis, emphysema, bronchial asthma and chronic obstructive pulmonary disease. It may also be used for paroxysmal dyspnoea associated with left heart failure.
Administration :
AMINOPHYLLINE INJECTION USP is administered by I.V. injection or infusion.
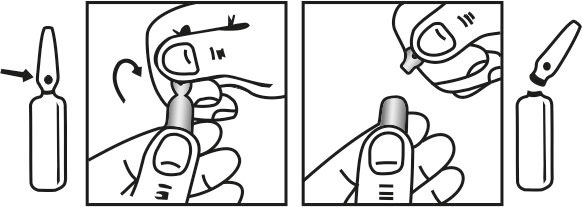
INSTRUCTIONS FOR USE OF AMPOULE :
The ampoule used in this product is equipped with O.P.C (One Point Cut) opening system. No ampoule file is needed to open the ampoule. The neck of the ampoule is prescored at the point of constriction. A coloured dot on the ampoule head helps to orientate the ampoule. Take the ampoule and face the coloured dot. Let the solution at the head of the ampoule to flow down by shaking or a gentle stroke. The ampoule opens easily by placing the thumb on the coloured dot and gently pressing downwards as shown:
Dosage :
Aminophylline Injection may be administered by intravenous infusion, or by slow intravenous injection at a rate not exceeding 20 to 25 mg/min. Recommended doses are given as a guide only. Dosage must be individualised based on patient characteristics, clinical response, and steady state theophylline concentration. Doses should be calculated on lean (ideal) body weight. Oral theophylline therapy should be substituted for intravenous therapy as soon as adequate improvement has been made. A loading dose is generally administered over 20 to 30 minutes, followed by a maintenance dose.
Adults and Children 6 months and over :
For patients not currently undergoing aminophylline or theophylline therapy, a dose of 6 mg aminophylline/kg lean body weight should be infused over 20 to 30 minutes, to provide a peak serum theophylline concentration of approximately 10 mcg/ml (55 micromole/l). For patients currently undergoing aminophylline or theophylline therapy, a serum theophylline concentration should be obtained. The dose of aminophylline may be administered on the principle that 0.6 mg aminophylline/kg lean body weight will increase the serum theophylline concentration by 1 mcg/ml. If it is not possible to obtain serum theophylline concentration, a dose of 3 mg aminophylline /kg lean body weight may be administered.
CONTRAINDICATIONS :
AMINOPHYLLINE INJECTION USP is contraindicated in
• In patients hypersensitive to xanthines or to ethylenediamine.
• In patients with coronary artery disease where myocardial stimulation might prove harmful.
• In patients with bronchiolitis (bronchopneumonia).
WARNINGS AND PRECAUTIONS :
AMINOPHYLLINE INJECTION USP should be used with extreme caution in patients currently undergoing therapy with other xanthines, such as theophylline, as the hazard of serious toxicity is increased. A serum theophylline concentration should always be obtained in these patients prior to any aminophylline administration.
AMINOPHYLLINE INJECTION USP should be used with caution in :-
• Elderly patients,
• Premature or neonatal infants,
• Patients with congestive heart failure,
• Chronic alcoholism,
• Acute febrile illness,
• Chronic obstructive pulmonary disease,
• Cor pulmonale,
• Influenza or those undergoing influenza immunization,
• Renal or hepatic dysfunction, including hepatic cirrhosis,
• Hypothyroidism,
• Acute pulmonary oedema or pneumonia,
Since clearance may be decreased and hence toxicity may be more likely in these patients. AMINOPHYLLINE INJECTION USP may lower the seizure threshold and should be administered with caution in patients with seizure disorder unless the patient is receiving appropriate anticonvulsant therapy. Dose adjustment of any anticonvulsant medication may be required.
AMINOPHYLLINE INJECTION USP should be administered with caution in patients with:-
• Peptic ulcer,
• Hyperthyroidism,
• Hypertension,
• Glaucoma,
• Diabetes mellitus,
• Tachyarrythmia,
• Gastroesophageal reflux,
Since these conditions may be exacerbated.
AMINOPHYLLINE INJECTION USP should be administered with caution in patients with :-
• Compromised cardiac or circulatory function,
• Angina pectoris or
• Acute myocardial injury
Where myocardial stimulation would be harmful.
Intravenous aminophylline must be administered slowly and cautiously to prevent dangerous CNS or cardiovascular toxicity. Too rapid intravenous administration may result in the following symptoms : anxiety, headache, nausea and vomiting, severe hypotension, dizziness, faintness, lightheadedness, palpitations, syncope, precordial pain, flushing, profound bradycardia, premature ventricular contractions, cardiac arrest. Intramuscular administration is not recommended as it causes intense local pain and sloughing of tissue. The coagulation time of the blood is shortened with aminophylline therapy.
Pregnancy : Pregnancy Category C.
There are no adequate and well controlled studies in pregnant women. Additionally, there are no teratogenicity studies in non-rodents (e.g., rabbits). Theophylline was not shown to be teratogenic in CD-1 mice at oral doses up to 400 mg/kg, approximately 2.0 times the human dose on a mg/m2 basis or in CD-1 rats at oral doses up to 260 mg/kg, approximately 3.0 times the recommended human dose on a mg/m2 basis. At a dose of 220 mg/kg, embryotoxicity was observed in rats in the absence of maternal toxicity.
Nursing mothers :
Theophylline is excreted into breast milk and may cause irritability or other signs of mild toxicity in nursing human infants. The concentration of theophylline in breast milk is about equivalent to the maternal serum concentration. An infant ingesting a liter of breast milk containing 10-20 mcg/ml of theophylline per day is likely to receive 10-20 mg of theophylline per day. Serious adverse effects in the infant are unlikely unless the mother has toxic serum theophylline concentrations.
Paediatric Use :
Theophylline is safe and effective for the approved indications in children. The constant infusion rate of intravenous theophylline must be selected with caution in children since the rate of theophylline clearance is highly variable across the age range of neonates to adolescents. Due to the immaturity of theophylline metabolic pathways in children under the age of one year, particular attention to dosage selection and frequent monitoring of serum theophylline concentrations are required when theophylline is prescribed to children in this age group.
Laboratory interactions :
Dipyridamole-assisted myocardial perfusion studies :
Aminophylline reverses the effects of dipyridamole on myocardial blood flow, thereby interfering with the test results. Dipyridamole-assisted myocardial perfusion studies should not be performed if therapy with aminophylline cannot be withheld for 36 hours prior to the test.
Uric acid serum determinations :
Aminophylline produces false-positive elevations of serum uric acid as measured by the Bittner or colorimetric methods, but not by the uricase method.
INTERACTIONS :
The following drugs may decrease aminophylline clearance resulting in increased serum levels and the potential for increased toxicity : alcohol, high dose allopurinol (> 600 mg/day), beta-blockers, cimetidine, oestrogen containing oral contraceptives, diltiazem, disulfuram, recombinant alpha-interferon, methotrexate, mexiletine, propranolol, tacrine, thiabendazole, thyroid hormones, ticlopidine, verapamil, and macrolide antibiotics and quinolones (including erythromycin, clarithromycin, ciprofloxacin and enoxacin). The following drugs may increase the clearance of aminophylline, and thereby decrease serum concentrations, possibly resulting in subtherapeutic dosing : aminoglutethimide barbiturates including phenobarbitone and primidone, carbamazepine, isoprenaline, phenytoin, rifampicin, St Johns wort (Hypericum perforatum), sulfinpyrazone, thioamines and tobacco and marijuana smoking.
In addition, the following drugs may interact with aminophylline :
Adenosine : Aminophylline may antagonise the cardiovascular effects of adenosine.
Beta-agonists : Concurrent use of aminophylline and beta-agonists may produce increased cardiotoxic effects. Also, aminophylline may potentiate the hypoglycaemia which may be associated with administration of beta-agonists.
Beta-blocking agents (including ophthalmic agents) : Concurrent use of aminophylline and beta-blockers may result in an inhibition of the bronchodilatory effects of aminophylline.
Benzodiazepines : Concurrent use of aminophylline and benzodiazepines may result in a reduction or reversal of the sedative effects of benzodiazepines.
Cardiac glycosides : Aminophylline may enhance the sensitivity of the myocardium to, and the toxic potential of cardiac glycosides.
Ephedrine and other sympathomimetic amines : Concurrent use of aminophylline and sympathomimetic amines may result in increased nausea, nervousness or insomnia.
Halothane : Concurrent use of aminophylline and halothane may result in ventricular arrythmias.
Ketamine : Concurrent use of aminophylline and ketamine may result in a lowered seizure threshold.
Lithium : Concurrent use of aminophyline and lithium may result in increased excretion of lithium, and hence a reduction in the therapeutic effect of lithium. Adjustment of the lithium dosage may be required.
Neuromuscular blocking agents, non-depolarizing : Aminophylline may antagonize the neuromuscular blocking effects of these agents.
Xanthines : Concurrent use of aminophylline and other xanthine containing medications may result in additive toxicity and should be avoided.
INCOMPATIBILITIES :
Incompatibility has been reported with chlorpromazine, clindamycin, corticotrophin, dimenhydrinate, doxorubicin, erythromycin gluceptate, hydralazine hydrochloride, hydroxyzine hydrochloride, opioid analgesics, oxytetracycline hydrochloride, phenytoin sodium, procaine hydrochloride, prochlorperazine salts, promazine hydrochloride, promethazine hydrochloride, sulphafurazole diethanolamine and vancomycin hydrochloride.
SIDE EFFECTS :
Cardiovascular system :
Tachycardia, palpitations, extrasystoles, increased pulse rate, flushing, hypotension, circulatory failure, atrial and ventricular arrhythmia, peripheral vasocontriction.
Central nervous system :
Headache, nervousness, insomnia, irritability, restlessness, dizziness, reflex hyperexcitability, seizures, anxiety, tremor, lightheadedness, excitement.
Gastrointestinal system :
Nausea, vomiting, heartburn, epigastric pain, abdominal cramps, anorexia, diarrhoea, haematemesis.
Genitourinary :
Increased urination, albuminuria.
Other :
Fever.
Respiratory system :
Tachypnoea.
Skin and appendages :
Ethylenediamine hypersensitivity induced dermatitis (hives, skin rash, sloughing of skin).
OVERDOSAGE :
Clinical features :
Less severe toxicities do not always precede major toxicities. Chronic overdose may produce toxicity at serum levels lower than those in acute overdose. Potentially life threatening toxicities may occur at serum concentration greater than 40 microgram/ml (220 micromole/L) in chronic overdose. In acuteoverdose serum concentrations greater than 90 microgram/mL (495 micromole/) are generally associated with severe toxicity.
TREATMENT OF OVERDOSAGE :
There is no specific antidote for aminophylline overdose. Treatment of overdose is symptomatic and supportive. Administration of sympathomimetic drugs should be avoided.
Treatment may involve the following measures :
• Administration of oral activated charcoal, regardless of the route of exposure to aminophylline (this assists in decreasing the serum concentration of theophylline by interrupting the enterohepatic circulation). Oral activated charcoal should be repeated until the serum theophylline concentration is below 20 microgram/ml.
• Charcoal haemoperfusion to increase the elimination of aminophylline. Haemodialysis is less effective in eliminating aminophylline, but may be warranted in some patients.
• Administration of intravenous diazepam to control seizures. Where diazepam is ineffective, phenytoin, phenobarbitone, or thiopentone may be considered.
• Correction of fluid and electrolyte balance.
• Support of respiratory functions by airway management, oxygen administration or mechanical ventilation as required.
• Support of cardiac functions. Propranolol may be warranted in the presence of extreme tachycardia, and antiarrythmic therapy may be required.
• Administration of phenothiazines in the presence of life threatening hypothermia.
• Monitoring of serum theophylline concentrations and ECG.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
STORAGE :
Store below 30°C (86°F), protected from light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
AMINOPHYLLINE INJECTION USP is supplied as 250 mg of aminophylline anhydrous USP in 10 ml aqueous solution.
Such 5 ampoules of 10 ml are packed in a box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.
 Cardiovascular
Cardiovascular