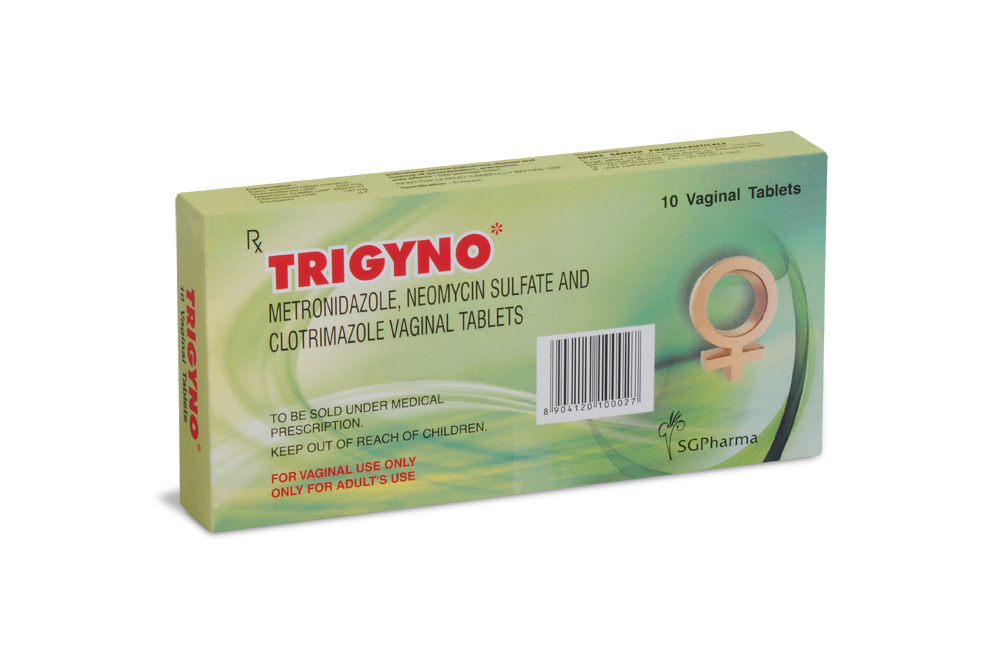
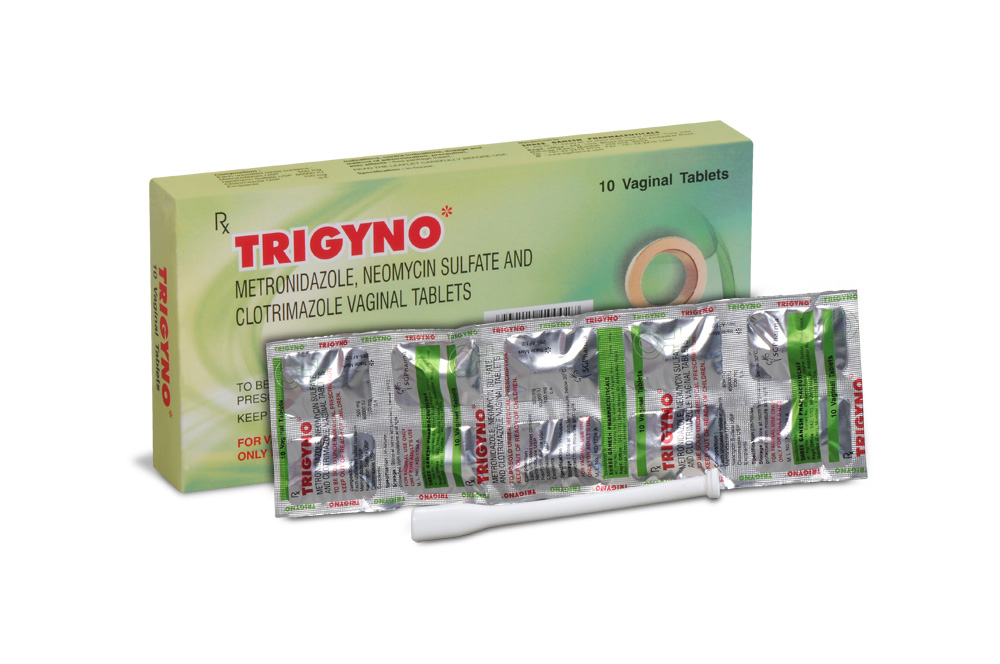
500 mg + 65,000 IU + 100 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
Cream coloured elongated uncoated tablets, having one end flat & depressed and the other end circular.
COMPOSITION :
Each uncoated tablet contains :
Metronidazole USP 500 mg
Neomycin Sulfate USP 65000 IU
Clotrimazole USP 100 mg
Excipients q.s.
(Corn Starch USP–NF, Microcrystalline Cellulose USP– NF,
Carboxymethylcellulose Sodium USP–NF, Purified Talc B.P., Methylparaben Sodium USP–NF, Propylparaben Sodium USP–NF)
SPECIFICATIONS :
Manufacturers (In-House).
ACTIONS :
TRIGYNO is a combination of antifungal, antibacterial & anti-infective drugs locally acting on urethra and vagina.
Metronidazole : is a 5-nitroimidazole derivative acting as an antiprotozoal and antibacterial agent such as anaerobic bacteria both Gram (-) and Gram (+), Trichomonas vaginalis, Gardrenella vaginalis.
Neomycin sulfate is an aminoglycoside antibiotic acting against aerobic bacteria Gram (-). So, it has a good treatment of Bacterial vaginosis due to banal pyogenic germs.
Clotrimazole is a broad spectrum imidazole antifungal with antimicrobial activity. It acts effectively against yeasts (Candida albicans and other candida species) and other fungi (Trichophyton, Epidermophyton, Microsporum), bacteria Gram (+) and protozoa (Trichomonas vaginalis, Gardrenella vaginalis, Staphylococcus aureus, Streptococcus pyogenes, Hemophilus vaginalis). The action mechanism of clotrimazole is to destroy fungal membrane by inhibiting 14a-demethylase and then inhibit the biosynthesis of ergosterol in fungi, a main component in the fungal membrane.
In clinical reality, vaginitis is often due to a combination of pathogens such as Trichomonas, Candida albicans, bacteria Gram (-) and Gram (+) that makes the treatment complicated. Therefore, it is required to have drugs of combination that have good treatment Trichomonas, fungi and bacteria at the same time. TRIGYNO can meet this requirement.
INDICATIONS :
TRIGYNO is active against sensitive aerobic bacteria both Gram (+) and Gram (-), sensitive anaerobic bacteria both Gram (+) and Gram (-), fungi, and protozoa. Therefore, it is indicated for the treatment of vaginitis due to one or a combination of the above pathogens such as Bacterial vaginitis due to banal pyogenic germs, vaginitis due to Trichomonas and Gardnerella vaginalis, Fungal (Monilial) vaginitis due to Candida albicans, vaginitis with desquamative leucorrhoea, mixed vaginitis due to Trichomonas & Yeast or a combination of the pathogens.
Administration :
By vaginal route. NOT FOR ORAL USE. ONLY FOR ADULTS.
Insert the tablet deeply in the vagina, 1 or 2 tablets daily. The treatment duration is at least 10 days, if necessary, some case up to 2-3 weeks. Patients should take the medicine before going to bed. Your partner should also be treated. Symptomatic relief may occur in a few days but continue full course of treatment as advised by the physician.
CONTRAINDICATIONS :
Contraindicated in the patients with hypersensitivity (allergy) to any component in the composition of the product. Take the medicine with disulfiram only if indicated by the physician. Please consult your physician in case of any doubt.
WARNINGS :
If irritation, sensitisation, pull brained or moving unregulation is found discontinue the treatment and consult your physician. Use of alcohol containing drinks should be reduced during the treatment with this drug (risk to reactions of red face, getting hot in the face, vomit and increase in heart rhythm). The patients should be instructed not to engage in vaginal intercourse during the treatment with this product. The treatment should not be carried out during menstruation but should be completed before this begins.
Pregnancy and lactation :
The drug is not indicated during pregnancy in the first trimester and lactation. If you are found to be pregnant in the first three months during the treatment of this medicine, please inform your physician for suitable treatment. However, pregnant women in the second and third trimester, lactating women can take this medicine if prescribed by doctor.
Ability to drive and operate machine :
Patient should be cautious because TRIGYNO may cause dizziness.
PRECAUTIONS :
In case of renal dysfunction or a history of serious nervous disease, please inform your physician. In case of history of leucocyte related hematological dysfunction, please consult your physician. Intractable candidiasis may be a symptom of unrecognized diabetes mellitus. Perform urine and blood glucose studies on patients who do not respond to treatment. A persistently resistant infection may actually be due to reinfection, evaluate a source of reinfection.
Laboratory test :
If there is lack of response, repeat microbiological studies to confirm diagnosis and rule out other pathogens before reinstituting antimycotic therapy.
INTERACTIONS WITH OTHER MEDICINES :
The medicine deactivates contraceptive method of spermicide. Metronidazole increases effects of some anti-blood coagulation drugs as warfarin, therefore, do not take those drugs together. In order to avoid interactions, especially disulfiram, please inform your physician about all medicines you are taking.
SIDE EFFECTS :
Rarely : Gastro intestinal dysfunction, vomit, nausea, diarrhoea, belly cramp, dizziness, headache, urticaria, pruritus, glossitis hot face, recoverable pancreatitis, confusion, convulsive seizures, vertigo, irritability, depression, weakness, incordination.A brown colouring of the urine frequently occurs.
Local side effects :
symptoms in the form of local prickling or irritation during the first days of treatment have been reported in rare instances. Inform the physician for any adverse effect.
OVERDOSAGES AND TREATMENT OF OVERDOSAGE :
High dose and/or long term therapy can cause decrease in leucocyte numbers. If taking by oral route a large amount of this medicine, it is advised to use stomach washing method if necessary. It is also advised to treat overdosage if taking by oral route from 12 g metronidazole up. There is no anti-dote and then symptoms treatments are applied. Overdosage symptoms are vomit, nausea, diarrhoea, stomachache, pruritus, convulsive seizures, decrease of leucocyte numbers, metal taste, incordination, brown colouring of the urine.
STORAGE :
Store at room temperature, below 30°C, protected from moisture and light. Do not store in the bathroom, near the kitchen sink, or in other damp places. Heat or moisture may break down the medicine.
SHELF LIFE :
36 months from date of manufacture.
PRESENTATION :
Pack of 10 vaginal tablets, packed in Aluminium foil.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.
 Cardiovascular
Cardiovascular