250 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
KOXSERINE (Cycloserine) is a broad–spectrum antibiotic that is produced by a strain of Streptomyces orchidaceus and has also been synthesized. Chemically, cycloserine is (+)-4-Amino-3-isoxazolidinone. The molecular formula is C3H6N2O2 and molecular weight is 102.09.
STRUCTURAL FORMULA :
Its structural formula is :
-Structure.jpg)
KOXSERINE tablets are white round film coated tablets.
COMPOSITION :
Each film coated tablet contains :
Cycloserine I.P. 250 mg
Excipients q.s.
Colour : Titanium Dioxide I.P.
ACTIONS :
Cycloserine, a broad-spectrum antibiotic, may be bactericidal or bacteriostatic, depending on its concentration at the site of infection and the susceptibility of the organism. Cycloserine is an analog of the amino acid D-alanine. It interferes with an early step in bacterial cell wall synthesis in the cytoplasm by competitive inhibition of 2 enzymes, L-alanine racemase, which forms D-alanine from L-alanine, and D-alanine- D-alanine synthetase, which incorporates D-alanine into the pentapeptide necessary for peptidoglycan formation and bacterial cell wall synthesis. Cycloserine has variable activity against Gram-positive and Gram-negative bacteria including Escherichia coli and Staphylococcus aureus. Cycloserine is active against Mycobacterium tuberculosis and some other mycobacteria. Resistance develops if cycloserine is used alone.
PHARMACOKINETICS :
Cycloserine is readily and almost completely absorbed from the gastrointestinal tract following oral administration. Peak plasma concentrations of 10 micrograms/ml have been obtained 3 to 4 hours after a dose of 250 mg, rising to 20 to 30 micrograms/ml on repeating the dose every 12 hours. The plasma half-life is about 10 hours and is prolonged in patients with renal impairment. Cycloserine is widely distributed into body tissues and fluids, including the CSF and breast milk. It crosses the placenta producing foetal blood concentrations approaching those in maternal serum. Cycloserine is excreted largely unchanged by glomerular filtration. About 50 % of a single 250 mg dose is excreted unchanged in the urine within 12 hours and about 70 % is excreted within 72 hours. As negligible amounts of cycloserine appear in the faeces, it is assumed that the remainder of a dose is metabolised to unidentified metabolites. It is removed by haemodialysis.
INDICATIONS :
KOXSERINE is indicated in the treatment of active pulmonary and extrapulmonary tuberculosis (including renal disease) when the causative organisms are susceptible to this drug and when treatment with the primary medications (streptomycin, isoniazid, rifampin and ethambutol) has proved inadequate. Like all antituberculosis drugs, KOXSERINE should be administered in conjunction with other effective chemotherapy and not as the sole therapeutic agent. KOXSERINE may be effective in the treatment of acute urinary tract infections caused by susceptible strains of gram–positive and gram–negative bacteria, especially Enterobacter spp. and Escherichia coli. It is generally no more and is usually less effective than other antimicrobial agents in the treatment of urinary tract infections caused by bacteria other than mycobacteria. Use of KOXSERINE in these infections should be considered only when more conventional therapy has failed and when the organism has been demonstrated to be susceptible to the drug.
Administration :
Oral.
Dosage :
KOXSERINE is effective orally and is currently administered only by this route. The usual dosage is 500 mg to 1 g daily in divided doses monitored by blood levels. The initial adult dosage most frequently given is 250 mg twice daily at 12–hour intervals for the first 2 weeks. A daily dosage of 1 g should not be exceeded.
Children :
The usual starting dose is 10 mg/kg/day, then adjusted according to blood levels obtained and therapeutic response.
Elderly :
As for adults but reduce dosage if renal function is impaired.
CONTRAINDICATIONS :
Administration is contraindicated in patients with any of the following :
Hypersensitivity to cycloserine
Epilepsy
Depression, severe anxiety or psychosis
Severe renal insufficiency
Excessive concurrent use of alcohol
WARNINGS :
Administration of KOXSERINE should be discontinued or the dosage reduced if the patient develops allergic dermatitis or symptoms of CNS toxicity such as convulsions, psychosis, somnolence, depression, confusion, hyperreflexia, headache, tremor, vertigo, paresis or dysarthria. The toxicity of KOXSERINE is closely related to excessive blood levels (above 30 μg/ml), as determined by high dosage or inadequate renal clearance. The ratio of toxic dose to effective dose in tuberculosis is small. The risk of convulsions is increased in chronic alcoholics. Patients should be monitored by haematologic, renal excretion, blood level and liver function studies.
PRECAUTIONS :
General :
Before treatment with KOXSERINE is initiated, cultures should be taken and the organism’s susceptibility to the drug should be established. In tuberculous infections, the organism’s susceptibility to the other antituberculosis agents in the regimen should also be demonstrated. Anticonvulsant drugs or sedatives may be effective in controlling symptoms of CNS toxicity, such as convulsions, anxiety and tremor. Patients receiving more than 500 mg of KOXSERINE daily should be closely observed for such symptoms. The value of pyridoxine in preventing CNS toxicity from KOXSERINE has not been proved. Administration of KOXSERINE and other antituberculosis drugs has been associated in a few instances with vitamin B12 and/or folic–acid deficiency, megaloblastic anaemia, and sideroblastic anaemia. If evidence of anaemia develops during treatment, appropriate studies and therapy should be instituted.
Laboratory Tests :
Blood levels should be determined at least weekly for patients with reduced renal function, for individuals receiving a daily dosage of more than 500 mg, and for those showing signs and symptoms suggestive of toxicity. The dosage should be adjusted to keep the blood level below 30 μg/ml.
Carcinogenesis, Mutagenicity, and Impairment of Fertility :
Studies have not been performed to determine potential for carcinogenicity. The Ames test and unscheduled DNA repair test were negative. A study in 2 generations of rats showed no impairment of fertility relative to controls for the first mating but somewhat lower fertility in the second mating.
Pregnancy : Category C
Cycloserine crosses the placenta; foetal serum concentrations may approach maternal serum concentrations. A study in 2 generations of rats given doses up to 100 mg per kg of body weight per day demonstrated no teratogenic effect in the offspring.
Nursing mothers :
Cycloserine is distributed into breast milk; concentrations may approach or exceed maternal serum concentrations.
Paediatric Use :
Appropriate studies on the relationship of age to the effects of cycloserine have not been performed in the paediatric population. However, no paediatrics-specific problems have been documented to date.
INTERACTIONS AND INCOMPATIBILITIES :
Concurrent administration of ethionamide has been reported to potentiate neurotoxic side effects. Alcohol and cycloserine are incompatible, especially during a regimen calling for large doses of the latter. Alcohol increases the possibility and risk of epileptic episodes. Patients receiving cycloserine and isoniazid should be monitored for signs of CNS toxicity, such as dizziness and drowsiness, as these drugs have a combined toxic action on the CNS. Dosage adjustments may be necessary.
SIDE EFFECTS :
The most frequent adverse effects with cycloserine involve the CNS and include anxiety, confusion, disorientation, depression, psychoses possibly with suicidal tendencies, aggression, irritability and paranoia. Vertigo, headache, drowsiness, speech difficulties, tremor, paresis, hyperreflexia, dysarthria, paraesthesia, coma and convulsions may also occur. Neurological reactions are dose related and may be reduced by keeping plasma concentrations below 30 micrograms/ml. It has been reported that up to 30 % of patients have experienced adverse effects. These reactions usually subside when cycloserine is discontinued or the dosage is reduced. Pyridoxine has been used in an attempt to treat or prevent neurological reactions but its value is unproven.
Hypersensitivity reactions including skin reactions and photosensitivity occur rarely. Serum aminotransferase values may be raised, especially in patients with a history of liver disease. Folate and vitamin B12 deficiency, megaloblastic anaemia, and sideroblastic anaemia have been reported occasionally when cycloserine has been used with other antituberculous drugs. Heart failure has occurred in patients receiving daily doses of 1 g or more.
INFORMATION FOR PATIENTS :
In providing consultation, consider emphasizing the following selected information (>> = major clinical significance) :
Before using this medication :
>> Conditions affecting use, especially :
Pregnancy : Cycloserine crosses the placenta and foetal serum concentrations may approach maternal serum concentrations.
Breast-feeding : Cycloserine is distributed into breast milk. Concentrations may approach or exceed maternal serum concentrations. Other medications, especially alcohol and ethionamide. Other medical problems, especially alcoholism (active or in remission), a history of seizure disorders, or renal function impairment.
Proper use of this medication :
Taking this medication after meals if gastrointestinal irritation occurs.
- Compliance with full course of therapy; in tuberculosis, therapy may take months or years.
- Importance of not missing doses and taking at evenly spaced times.
- Proper dosing.
Missed dose: Taking as soon as possible; not taking if almost time for next dose; not doubling doses.
- Proper storage.
Precautions while using this medication :
Regular visits to physician to check progress. Checking with physician if no improvement within 2 to 3 weeks.
- Checking with physician immediately if thoughts of suicide occur.
- Caution if dizziness or drowsiness occurs.
- Avoiding alcoholic beverages while taking this medication.
OVERDOSAGE :
Signs and Symptoms :
Acute toxicity from cycloserine can occur if more than 1 g is ingested by an adult. Chronic toxicity from cycloserine is dose related and can occur if more than 500 mg is administered daily. Patients with renal impairment will accumulate cycloserine and may develop toxicity if the dosing regimen is not modified. Patients with severe renal impairment should not receive the drug. The central nervous system is the most common organ system involved with toxicity. Toxic effects may include headache, vertigo, confusion, drowsiness, hyperirritability, paresthesias, dysarthria and psychosis. Following larger ingestions, paresis, convulsions and coma often occur. Ethyl alcohol may increase the risk of seizures in patients receiving cycloserine. The oral median lethal dose in mice is 5290 mg/kg.
TREATMENT OF OVERDOSAGE :
The following is provided to serve as a guide should such an overdose be encountered. Protect the patient’s airway and support ventilation and perfusion. Meticulously monitor and maintain, within acceptable limits, the patient’s vital signs, blood gases, serum electrolytes etc. Absorption of drugs from the gastrointestinal tract may be decreased by giving activated charcoal, which, in many cases, is more effective than emesis or lavage; consider charcoal instead of or in addition to gastric emptying. Repeated doses of charcoal over time may hasten elimination of some drugs that have been absorbed. Safeguard the patient’s airway when employing gastric emptying or charcoal. In adults, many of the neurotoxic effects of cycloserine can be both treated and prevented with the administration of 200 to 300 mg of pyridoxine daily. The use of haemodialysis has been shown to remove cycloserine from the bloodstream. This procedure should be reserved for patients with life-threatening toxicity that is unresponsive to less invasive therapy.
STORAGE :
Store below 250C (770F), protected from moisture and light.
SHELF LIFE :
24 months from the date of manufacture.
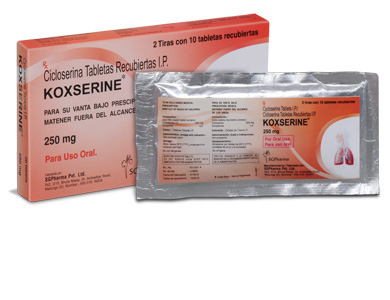
PRESENTATION :
KOXSERINE tablets contain Cycloserine I.P. 250 mg.
2 Strips of 10 tablets per box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.
 Cardiovascular
Cardiovascular